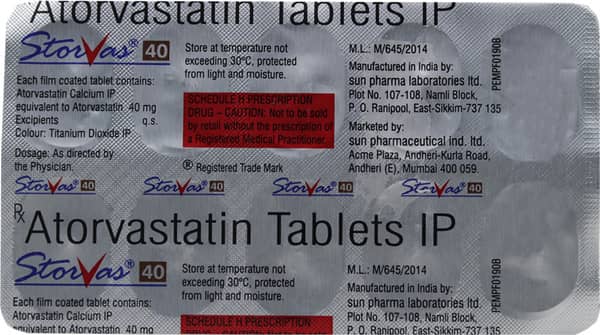
Storvas 40 Tablet
10 Tablet(s) in StripMRP ₹206.05
₹154.54
25% OFF
Made by
RANBAXY LABORATORIES (SUN PHARMA)
Dosage
TABLET
Expiry
Product Summary
| Salt Content | Atorvastatin(40.0 Mg) |
| Uses | Lipid-lowering |
| Therapy | Hypolipidemic drugs |
| Side effects | Headache, hyperglycaemia, constipation, nausea |
Assurance
available
Regulated
Medical Description
Storvas 40 mg Tablet contains atorvastatin, which is a medicine used to lower high blood cholesterol levels. It is used in people whose diet and lifestyle changes have not been enough to lower their cholesterol levels. Storvas 40 mg Tablet works by blocking an enzyme that your body uses to make cholesterol. This lowers the amount of cholesterol in your blood.
High blood cholesterol is a condition in which there is too much bad cholesterol and triglycerides in the blood. This can lead to the buildup of plaque in the arteries, which can narrow or block them. This can lead to heart attack, stroke and other health problems.
Storvas 40 mg Tablet can be used by adults and children aged 10 years and over. Along with taking Storvas 40 mg Tablet, you should also follow a cholesterol-lowering diet, make lifestyle changes and exercise regularly. Aztor 40mg tablet, Tonact 40mg tablet, Storvas 40mg tablet and Lipvas 40mg tablet are some other medicines with atorvastatin as their active medicines.
Do not consume alcohol or grapefruit juice while taking Storvas 40 mg Tablet. These can make Storvas 40 mg Tablet less effective.
Before taking Storvas 40 mg Tablet, tell your doctor about all the other medicines you are taking, including over-the-counter medicines, vitamins and herbal supplements. Storvas 40 mg Tablet can interact with other medicines and cause side effects. Do not take Storvas 40 mg Tablet if you are pregnant or breastfeeding.
Uses
Contraindications
- If you are allergic to Atorvastatin or any other components of Storvas Tablet.
- If you have or ever had any liver disease, any abnormality in the liver function test.
- If you are being treated with medicines for hepatitis C like glecaprevir/pibrentasvir.
- If you are pregnant or are breastfeeding.
Side effects
- Headache
- Nausea
- Diarrhoea
- Body pain (joint, back, throat, muscle)
- Swelling of nasal passages and bleeding from the nose
- An abnormal liver function test and increased creatinine level
Precautions and Warnings
Pregnancy
Breast Feeding
Driving
Alcohol
Other General Warnings
- You have liver or kidney diseases.
- You experienced any muscle problems after taking other lipid-lowering medicines.
- You are taking medicines for bacterial infection like fusidic acid, as this may cause serious muscle injury.
- You have respiratory failure.
- You are addicted to alcohol.
- You have any thyroid problems.
- You have an inability to digest lactose (lactose intolerance).
Directions for Use
Storage and disposal
- Store it at a temperature below 30°C in a cool and dry place.
- Keep it out of reach of children and pets.
- Check its expiry date before starting the treatment.
Dosage
Overdose
Missed a Dose
Mode of Action
How Does It Work?
Interactions
Interactions with other medicines
- HIV medications such as ritonavir with Storvas Tablet can result in severe side effects.
- Erythromycin (antibiotic) taken with Storvas Tablet can result in increased side effects.
- It should be ensured that fusidic acid is taken in a proper gap with this medicine, a minimum gap of seven days is advised.
- Discuss with your doctor about any other medicines, herbal preparations and supplements you are taking currently to avoid any possible interactions.
Interactions with food items
Frequently Asked Questions (FAQs)
Articles
View AllChronic Condition Articles
View AllDid you find this helpful?
Explore More at Pharmeasy
Manufacturer Details
Brand:STORVAS
Manufacturer Name:RANBAXY LABORATORIES (SUN PHARMA)
Address:Sun Pharma Laboratories Ltd.,Plot No107,108 Mamli Block,P.O. Ranipool,East Sikkim 737135
Country of Origin:India
Vendor Details
Fulfillment Information: Storvas 40 MG Tablet (10) will be delivered by a licensed retail pharmacy nearest to your location. Order acceptance and fulfillment are dependent upon the verification of your doctor's valid prescription (where applicable) and the current stock of this medicine.
References:
- EMA. SmPC. Atorvastatin. [ Accessed on 20.March.2021 ] (online)
- CDSCO. Atorvastatin. [ Accessed on 20.March.2021 ](online)
- National Center for Biotechnology Information (2024). PubChem Compound Summary for CID 60823, Atorvastatin. Retrieved January 28, 2025.
- ScienceDirect. Atorvastatin. Accessed January 28, 2025.
- Atorvastatin. Labeling information. U.S. Food and Drug Administration [Internet]. 2009 [cited 2025 Jan 28].
In Case of Any Issues Contact Us
Email Id:care@pharmeasy.in
Phone Number:7666100300
Address:D-37/1, Opp. Sandoz, TTC Industrial Area, MIDC Industrial Area, Turbhe, Navi Mumbai, Maharashtra 400703
Disclaimer
The information provided herein is supplied to the best of our abilities to make it accurate and reliable as it is published after a review by a team of professionals. This information is solely intended to provide a general overview on the product and must be used for informational purposes only. You should not use the information provided herein to diagnose, prevent, or cure a health problem. Nothing contained on this page is intended to create a doctor-patient relationship, replace or be a substitute for a registered medical practitioner's medical treatment/advice or consultation. The absence of any information or warning to any medicine shall not be considered and assumed as an implied assurance. We highly recommend that you consult your registered medical practitioner for all queries or doubts related to your medical condition. You hereby agree that you shall not make any health or medical-related decision based in whole or in part on anything contained in the Site. Please click here for detailed T&C.