Rotasiil-Liquid Oral Vaccine
2ml Oral Vaccine in AmpouleMRP ₹924.53
₹647.17
30% OFF
Made by
SERUM INSTITUTE OF INDIA PRIVATE LIMITED
Dosage
ORAL VACCINE
Expiry
Product Summary
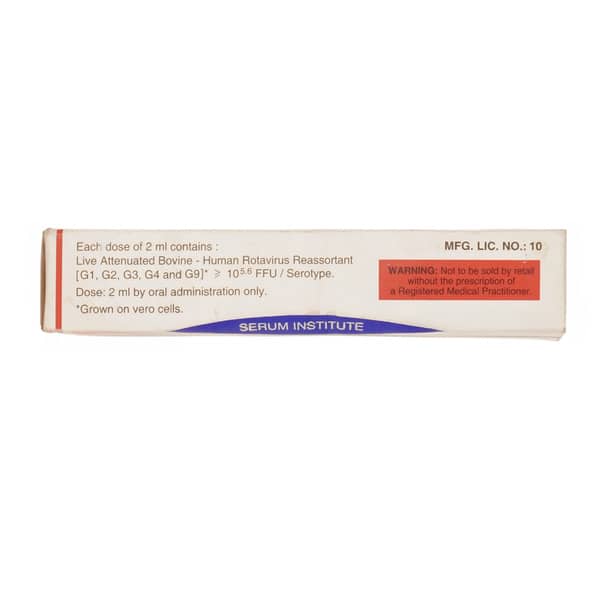
| Salt Content | Rotavirus Vaccine Live Attenuated(100000.0 Iu) |
| Uses | Vaccine for rotavirus |
| Therapy | Vaccine |
| Side effects | mild irritability, abdominal pain, flatulence |
Assurance
available
Regulated
Medical Description
Rotasiil-Liquid Oral Vaccine is a specialized medicine designed to protect infants from rotavirus, a highly contagious virus that is a leading cause of severe "stomach flu" or gastroenteritis. Unlike many other immunizations that require a needle, this protection is delivered as a liquid given by mouth, making the process much easier for your baby. It works by introducing a live but significantly weakened version of the virus to the digestive system, which teaches your child’s immune system how to recognize and fight off the actual disease if they are exposed to it later in life.
Because the vaccine is administered orally, it specifically targets the area where rotavirus typically attacks. Rotasiil-Liquid Oral Vaccine is generally given in a series of doses during the first few months of an infant's life to ensure their natural defenses are strong before they are likely to encounter the virus in daycare or community settings. It is a vital tool in preventing the extreme dehydration and hospitalizations that often occur when babies suffer from the intense vomiting and diarrhea associated with a rotavirus infection.
While Rotasiil-Liquid Oral Vaccine is safe for most infants, some children may experience mild and temporary reactions as their bodies respond to the vaccine. Common signs to look out for include slight irritability, a stuffy or runny nose (nasopharyngitis), or mild vomiting. In some cases, parents might notice symptoms like ear inflammation or a temporary change in breathing patterns. Most of these side effects are short-lived and resolve on their own as the baby adjusts to the medicine.
It is important to keep a close eye on your child after they receive Rotasiil-Liquid Oral Vaccine. If any of these side effects seem to linger, become more severe, or if your baby seems unusually distressed, you should reach out to your pediatrician or healthcare provider. They can offer personalized advice on managing these symptoms at home or determine whether any further care is needed. By staying observant and following the recommended schedule, you are taking a major step in keeping your baby healthy and hydrated.
Uses
Contraindications
- Are allergic to the active ingredient (live attenuated rotavirus vaccine) or any of the other ingredients in the vaccine (like sucrose, sodium, glucose, or phenylalanine).
- Have had an allergic reaction to a rotavirus vaccine before.
- Have a history of intussusception (a serious condition where one part of the intestine slides into another).
- Have an uncorrected birth defect of the digestive tract that could lead to intussusception.
- Have Severe Combined Immunodeficiency (SCID), a serious condition affecting the immune system.
- Are suffering from a severe feverish illness. However, a minor infection is usually not a reason to postpone vaccination.
- Are currently suffering from diarrhoea or vomiting.
Side effects
- Diarrhea
- Irritability
- Abdominal (tummy) pain
- Flatulence (gas)
- Dermatitis (skin rash or inflammation)
Precautions and Warnings
Other General Warnings
- You or your baby has a history of intussusception (a serious intestinal condition).
- You or your baby has an uncorrected congenital malformation of the gastrointestinal tract that would predispose to intussusception.
- You or your baby has Severe Combined Immunodeficiency (SCID) disorder.
- You or your baby is suffering from an acute severe febrile illness (fever).
- You or your baby is suffering from diarrhoea or vomiting.
- You have a known or suspected immunodeficiency, including in utero exposure to an immunosuppressive treatment (for your baby).
- You have immunodeficient close contacts (such as individuals with malignancies, or who are otherwise immunocompromised, or individuals receiving immunosuppressive therapy).
- You have a very premature infant (born at or before 28 weeks of gestation) with a previous history of respiratory immaturity.
- You or your baby has a rare hereditary problem, such as fructose intolerance, glucose-galactose malabsorption, or sucrase-isomaltase insufficiency.
- You or your baby has phenylketonuria (PKU).
Directions for Use
- The first dose can be given from 6 weeks of age.
- There should be at least 4 weeks between doses.
- The vaccination course should preferably be completed by 16 weeks of age, but must be finished by 24 weeks of age.
- If your baby spits out or throws up most of the vaccine, a single replacement dose can be given during the same visit.
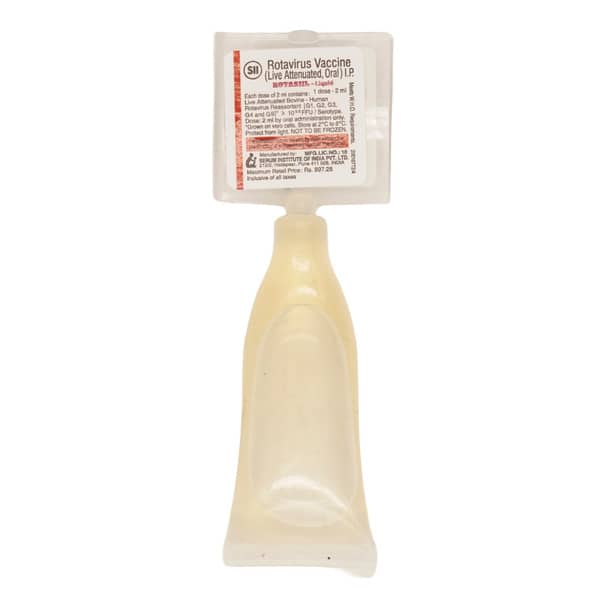
- Check the expiry date and ensure the tube isn't damaged or already open.
- Make sure the liquid is clear and colourless, with no particles. Do not use it if it looks abnormal.
- Pull off the cap and keep it.
- Flick the top of the tube repeatedly until liquid clears from the thinnest section just below the membrane.
- Hold the tube upright, turn the cap upside down (180 degrees), and press it down firmly to pierce the membrane. Do not twist. Then lift off the cap.
- Check that a hole has been made at the top of the tube. If not, repeat steps 2-4.
- Once the tube is open, give the vaccine immediately.
- Seat your child leaning slightly backwards.
- Gently squeeze the liquid into the side of your child's mouth, towards the inside of their cheek. You might need to squeeze a few times to get all the vaccine out. It's okay if a drop remains in the tip.
Storage and disposal
- Store Rotasiil oral vaccine in a refrigerator (2°C – 8°C).
- Do not freeze the vaccine.
- Keep it in its original package to protect it from light.
- The vaccine should be used immediately after opening.
- Any unused vaccine or waste materials should be disposed of in accordance with local regulations for the disposal of biological waste.
Quick Tips
- Rotasiil oral vaccine is for oral use only. It must never be injected.
- Ensure the vaccine is stored correctly (refrigerated, not frozen, protected from light).
- Always inspect the vaccine for any foreign particles or unusual appearance before administration. If observed, discard the vaccine.
Dosage
Overdose
Missed a Dose
Mode of Action
How Does It Work?
Interactions
Interactions with other medicines
- Rotasiil oral vaccine can be given at the same time as other routine childhood vaccines, such as those for diphtheria, tetanus, pertussis, Haemophilus influenzae type b, polio, hepatitis B, pneumococcal disease, and meningococcal disease. These vaccines generally do not affect each other's effectiveness or safety.
- Even if Rotasiil oral vaccine is given with oral polio vaccine (OPV), it doesn't affect the polio protection. While OPV might slightly reduce the immune response to the rotavirus vaccine, the protection against severe rotavirus stomach flu is maintained.
- There are no restrictions on your baby's food or liquid intake before or after vaccination.
Frequently Asked Questions (FAQs)
Articles
View AllChronic Condition Articles
View AllDid you find this helpful?
Explore More at Pharmeasy
Manufacturer Details
Brand:ROTASIIL
Country of Origin:India
Vendor Details
Fulfillment Information: Rotasiil 100000 IU Oral Vaccine (2) will be delivered by a licensed retail pharmacy nearest to your location. Order acceptance and fulfillment are dependent upon the verification of your doctor's valid prescription (where applicable) and the current stock of this medicine.
References:
In Case of Any Issues Contact Us
Email Id:care@pharmeasy.in
Phone Number:7666100300
Address:D-37/1, Opp. Sandoz, TTC Industrial Area, MIDC Industrial Area, Turbhe, Navi Mumbai, Maharashtra 400703
Disclaimer
The information provided herein is supplied to the best of our abilities to make it accurate and reliable as it is published after a review by a team of professionals. This information is solely intended to provide a general overview on the product and must be used for informational purposes only. You should not use the information provided herein to diagnose, prevent, or cure a health problem. Nothing contained on this page is intended to create a doctor-patient relationship, replace or be a substitute for a registered medical practitioner's medical treatment/advice or consultation. The absence of any information or warning to any medicine shall not be considered and assumed as an implied assurance. We highly recommend that you consult your registered medical practitioner for all queries or doubts related to your medical condition. You hereby agree that you shall not make any health or medical-related decision based in whole or in part on anything contained in the Site. Please click here for detailed T&C.