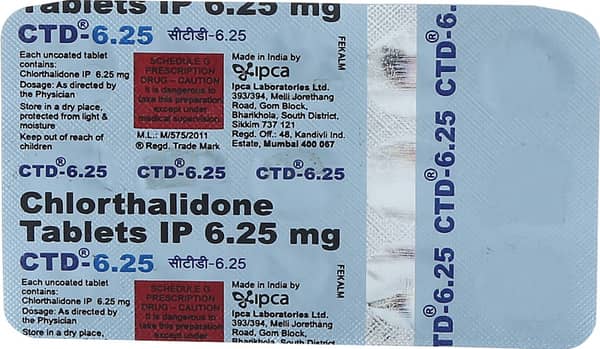
CTD 6.25 Tablet
15 Tablet(s) in StripMRP ₹128.21
₹98.72
23% OFF
Made by
IPCA LABORATORIES
Dosage
TABLET
Expiry
Product Summary
| Salt Content | Chlorthalidone / Chlortalidone(6.25 Mg) |
| Uses | Hypertension, edema |
| Therapy | Anti-hypertensive |
| Side effects | Headache, nausea, dizziness, low blood pressure |
Assurance
available
Regulated
Medical Description
CTD 6.25 is a medication used to treat high blood pressure. It contains chlorthalidone, a diuretic that helps lower blood pressure by increasing the amount of water and sodium your body removes through urine.
Take CTD 6.25 exactly as prescribed by your doctor, usually at the same time each day with food. Do not stop taking this medication suddenly, as it can cause serious health problems.
Common side effects include headache, nausea, and dizziness. Consult your doctor if these side effects bother you. Before taking CTD 6.25, inform your doctor about any existing kidney or liver problems. Pregnant or breastfeeding women should also consult their doctor. Tell your doctor about all other medications you are currently taking, as they may interact with CTD 6.25
New Ch tablet, Thaloric tablet and Lupiclor tablet also contain chlorthalidone as their main ingredient. Before using this medicine, inform the doctor if you are pregnant, planning a pregnancy or breastfeeding and about your detailed medical history.
Uses
Contraindications
- If you are allergic to Chlorthalidone or any of the other ingredients of this medicine.
- If your urine output in 24 hours is very low (less than 100ml).
Side effects
- Headache
- Nausea
- Dizziness
- Low blood pressure
- Weakness
Precautions and Warnings
Pregnancy
Breast Feeding
Driving
Alcohol
Other General Warnings
- Your blood pressure is low or you have dehydration.
- You have kidney disease or you are on a salt-restricted diet.
- The use of CTD 6.25 tablets may cause low levels of electrolytes in the body. It is advisable to monitor electrolytes periodically.
Directions for Use
Storage and disposal
- Store CTD 6.25 tablet at room temperature in a cool and dry place.
- Keep it out of the reach of children and pets.
Quick Tips
- Some individuals may experience headaches, stomach pain, diarrhoea, itchiness, and nausea/vomiting while using this medicine. Inform your doctor if these effects bother you or do not go away.
- It can result in dehydration, salt imbalance or electrolyte imbalance.
- Your diet should avoid excessive salt and potassium. You should also exercise regularly and avoid alcohol and smoking while taking this medication.
- Some individuals may experience loss of appetite, stomach pain, fatigue, and nausea/vomiting while taking this medicine. Inform your doctor if these effects bother you or do not go away.
- If you experience dizziness or sleepiness after taking the CTD tablet, avoid driving, operating heavy machinery, or performing any tasks that require mental alertness.
Dosage
Overdose
Missed a Dose
Mode of Action
How Does It Work?
Interactions
Interactions with other medicines
- Some medicines can affect the way CTD 6.25 tablet works, or this medicine itself can reduce the effectiveness of other medicines taken at the same time.
- Tell your doctor about all the medicines, supplements, or herbals you are currently taking or might take to avoid any possible interaction.
- Taking CTD 6.25 tablets, along with other medications used for high blood pressure, increases the action of other medicines.
- Taking this medicine with lithium decreases the excretion of lithium through urine, leading to the accumulation of lithium in the body.
Interactions with food items
Frequently Asked Questions (FAQs)
Articles
View AllChronic Condition Articles
View AllDid you find this helpful?
Explore More at Pharmeasy
Manufacturer Details
Brand:CTD
Manufacturer Name:IPCA LABORATORIES
Address:Ipca Laboratories Ltd.,393/394, Melli. Jorethang Road,,Gom Block, Bharikhola,,South District, Sikkim-737121
Country of Origin:India
Vendor Details
Fulfillment Information: Ctd 6.25 MG Tablet (15) will be delivered by a licensed retail pharmacy nearest to your location. Order acceptance and fulfillment are dependent upon the verification of your doctor's valid prescription (where applicable) and the current stock of this medicine.
References:
- THALITONE [Internet]. Accessdata.fda.gov. 2025 [cited 17th Oct 2025]
- CDSCO [Internet]. Cdscoonline.gov.in. 2025 [cited 17th Oct 2025]
- National Center for Biotechnology Information (2025). PubChem Compound Summary for CID 2732, Chlorthalidone. 2025 [cited 17th Oct 2025]
- DrugBank. Chlorthalidone. 2025 [cited 17th Oct 2025]
- ScienceDirect. Chlortalidone. ScienceDirectTopics; 2025 [cited 17th Oct 2025]
- U.S. Food and Drug Administration. THALITONE® (chlorthalidone) Tablets USP 15 mg and 25 mg. FDA; 2018 2025 [cited 17th Oct 2025]
In Case of Any Issues Contact Us
Email Id:care@pharmeasy.in
Phone Number:7666100300
Address:D-37/1, Opp. Sandoz, TTC Industrial Area, MIDC Industrial Area, Turbhe, Navi Mumbai, Maharashtra 400703
Disclaimer
The information provided herein is supplied to the best of our abilities to make it accurate and reliable as it is published after a review by a team of professionals. This information is solely intended to provide a general overview on the product and must be used for informational purposes only. You should not use the information provided herein to diagnose, prevent, or cure a health problem. Nothing contained on this page is intended to create a doctor-patient relationship, replace or be a substitute for a registered medical practitioner's medical treatment/advice or consultation. The absence of any information or warning to any medicine shall not be considered and assumed as an implied assurance. We highly recommend that you consult your registered medical practitioner for all queries or doubts related to your medical condition. You hereby agree that you shall not make any health or medical-related decision based in whole or in part on anything contained in the Site. Please click here for detailed T&C.