Azarga Bottle Of 5Ml Eye Drops
5ml Eye Drop in BottleMRP ₹521.56
₹417.25
20% OFF
Made by
J B CHEMICALS AND PHARMACEUTICALS
Dosage
EYE DROP
Expiry
Product Summary
| Uses | Ocular hypertension, glaucoma |
| Therapy | Glaucoma drugs |
| Side effects | Blurred vision, eye pain, eye irritation, dry eye, itchy eye |
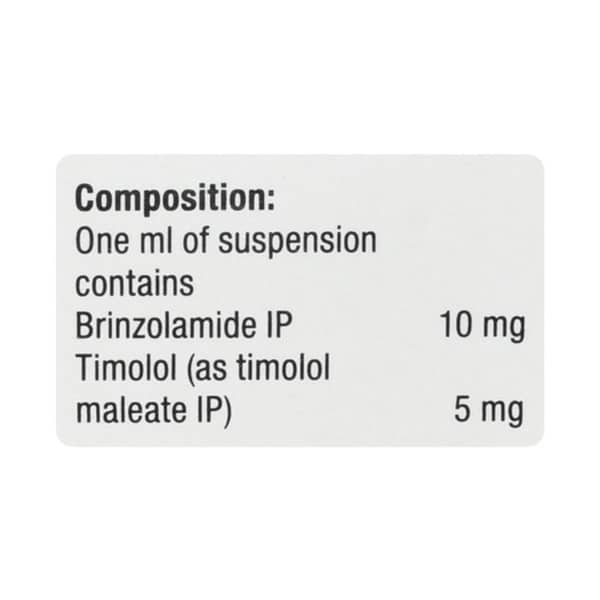
| Salt Content | Brinzolamide(10.0 Mg)+Timolol(5.0 Mg) |
Assurance
available
Regulated
Medical Description
Uses
- Ocular hypertension
- Glaucoma
Contraindications
- Known allergy to any ingredients of Azarga eye drops
- Known allergy to beta-blockers or sulphonamides class of medications
- A history of or currently suffering form bronchial asthma or severe chronic obstructive pulmonary disease
- Sick sinus syndrome
- Sinus bradycardia
- Sino-atrial block
- Atrioventricular block
- Severe allergic rhinitis
- Kidney problems
Side effects
- Blurred vision
- Eye pain
- Eye irritation
- Itchy eye
- Redness of the eye
Precautions and Warnings
Pregnancy
Breast Feeding
Driving
Alcohol
Other General Warnings
- You have heart problems such as coronary heart disease, heart failure, low blood pressure, and disturbances of heart rate such as slow heartbeat
- You have breathing problems, asthma, or chronic obstructive pulmonary disease
- You have poor blood circulation disease (such as Raynaud’s disease or Raynaud’s syndrome)
- You suffer from diabetes
- You have thyroid problems
- You have myasthenia gravis
- You have liver problems
- You have dry eyes or a cornea problem
- You have problems with your kidneys
- You suffer from dry eyes
Directions for Use
- Wash your hands before using the eye drops
- Shake well before use
- For external use only
- Remove the bottle cap by twisting it off
- Do not use the dropper to touch your eye, eyelid, adjacent areas, or other surfaces. It has the potential to infect the droplets.
- Use azarga eye drops as suggested by your doctor
Storage and disposal
- Keep this medication out of children's sight and reach
- After the expiry date, which is printed on the bottle and carton after EXP, do not use this medication
- To avoid infections, discard the bottle four weeks after it was first opened and replace it with a fresh one
- Do not dispose of medications in the wastewater or household waste
Dosage
Overdose
Missed a Dose
Mode of Action
How Does It Work?
Interactions
Interactions with other medicines
- CYP3A4 inhibitors such as ketoconazole, itraconazole, clotrimazole, ritonavir, and troleandomycin stop CYP3A4 enzyme from breaking down brinzolamide
- When an ophthalmic beta-blocker solution is used in conjunction with oral calcium channel blockers, beta-adrenergic blocking agents, antiarrhythmics (including amiodarone), digitalis glycosides, parasympathomimetic, and guanethidine, there is a risk of adverse effects like low blood pressure and heart rate
- The contents of azarga eye drops can reduce the effect of adrenaline
- Combined treatment with CYP2D6 inhibitors (e.g. quinidine, fluoxetine, paroxetine) and timolol cause a reduction heart rate or depression
- Concomitant use of antidiabetic medicine reduces the blood sugar-lowering effect of antidiabetics
- Concomitant use of adrenaline can cause dilated pupils
Frequently Asked Questions (FAQs)
Articles
View AllChronic Condition Articles
View AllDid you find this helpful?
Explore More at Pharmeasy
Manufacturer Details
Brand:AZARGA
Manufacturer Name:J B CHEMICALS AND PHARMACEUTICALS
Address:S.A. Alcon-Couvreur N.V., B-2870 Puurs, Belgium.
Country of Origin:India
Vendor Details
Fulfillment Information: Azarga Bottle Of 5ml Eye Drops will be delivered by a licensed retail pharmacy nearest to your location. Order acceptance and fulfillment are dependent upon the verification of your doctor's valid prescription (where applicable) and the current stock of this medicine.
References:
In Case of Any Issues Contact Us
Email Id:care@pharmeasy.in
Phone Number:7666100300
Address:D-37/1, Opp. Sandoz, TTC Industrial Area, MIDC Industrial Area, Turbhe, Navi Mumbai, Maharashtra 400703
Disclaimer
The information provided herein is supplied to the best of our abilities to make it accurate and reliable as it is published after a review by a team of professionals. This information is solely intended to provide a general overview on the product and must be used for informational purposes only. You should not use the information provided herein to diagnose, prevent, or cure a health problem. Nothing contained on this page is intended to create a doctor-patient relationship, replace or be a substitute for a registered medical practitioner's medical treatment/advice or consultation. The absence of any information or warning to any medicine shall not be considered and assumed as an implied assurance. We highly recommend that you consult your registered medical practitioner for all queries or doubts related to your medical condition. You hereby agree that you shall not make any health or medical-related decision based in whole or in part on anything contained in the Site. Please click here for detailed T&C.