A Complete Guide On MyLab’s CoviSelf Test Kit
By Deesha Jain +2 more

Get,

to manage your symptom
Get your,


4 Cr+ families
benefitted

OTP sent to 9988776655



You’ve successfully subscribed to receive
doctor-approved tips on
Whatsapp

Get ready to feel your best.

Hi There,



Register to Avail the Offer
Send OTPBy continuing, you agree with our Privacy Policy and Terms and Conditions

Hi There,

Trusted by 4 crore+ families

OTP sent to 9988776655



You have unlocked 25% off on medicines




Code: NU25

By Deesha Jain +2 more
Table of Contents
As the second wave of COVID-19 shook the roots of Indian healthcare machinery, some researchers used this disastrous time as an opportunity to prepare a self-testing kit for common people, which they can use to know if they are infected with SARS-CoV-2 or not within the safety and comfort of home in a very short time. The quick results given by CoviSelf will help you seek proper treatment at the earliest, thereby reducing the risk of developing severe illness due to delay in diagnosis and the initiation of treatment.
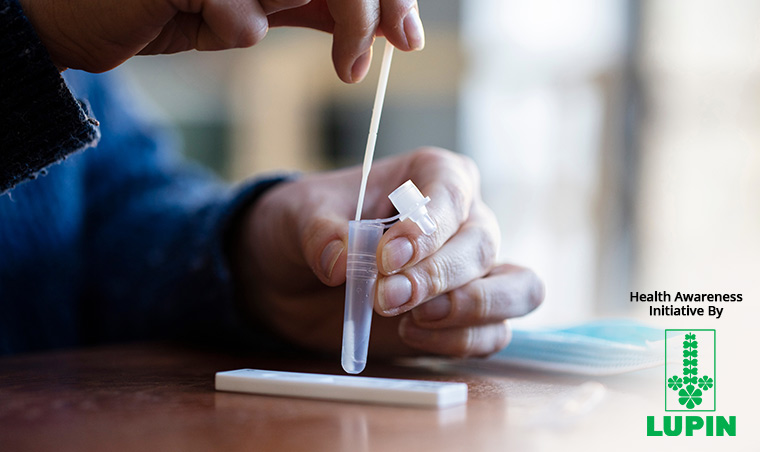
The self-testing kits, which can potentially release a huge burden off the healthcare system, have now also received a green signal from the Indian Council of Medical Research (ICMR).
The self-test kit, approved by ICMR, has been developed by a Pune-based molecular company named MyLab Discovery Solutions. The name of the kit is CoviSelf.
It uses a rapid antigen test. In it, the patient just has to take the nasal swab sample, and it will give the result within 15 minutes. It only takes about 2 minutes to carry out the test.
This test kit costs only Rs 250. The manufacturing company currently has a production capacity of 70 lakh kits per week, and they are expecting to expand it to one crore kits per week in the next 14 days. The kit will not only be sold at the local medical stores but also at the e-pharmacies in India.
Aiming to fight against the upcoming waves of COVID-19 with more efficiency, MyLab has also prepared an AI-powered smartphone app to update users about their positive status. They can also submit their report directly to the ICMR for traceability purposes. The app will also tell them what to do next in case of either result.
ICMR recommends taking this test only if you have any symptoms of COVID-19 or have come in contact with a COVID positive patient. If found positive, the patient will not require an RT-PCR test for confirmation, and all the government-specified protocols will be followed for isolation and high-risk contact tracing.
If your result comes out positive, your report will be automatically submitted on the ICMR portal, as the test is in sync with MyLab’s mobile app CoviSelf. ICMR, however, recommends not to take this test in public places.
Moreover, if a person has tested negative but still has symptoms, he/she is recommended to take an RT-PCR test.
The self-test kit includes the following:
To begin, the user has to download the CoviSelf app on their smartphone and enter all their details. This data will be saved on a secured server that is directly connected with the ICMR portal, making your test reports available to the government.
All the self test kits are rapid antigen detection kits which have high false negative rates. The definitive diagnosis should always be made by RTPCR.
Dr. M.G. Kartheeka, MBBS, MD
Covid self tests would be helpful ideally 5 days after an exposure to covid virus. If you are negative for the first time you may repeat the test after 48 hours for a total of 3 times.
Dr. Ashish Bajaj, M.B.B.S., M.D.
You will get the result within 15 minutes of taking the test. In case the result is positive, two lines will appear on the testing card. One line will be on marker “t” for the testing line, and “c” for the quality control line. If the result is negative, there will be only one line at marker “c”.
If the result takes more than 20 minutes to appear or if you can not see any line on marker “c”, then the test is considered invalid.
Many states in India are currently struggling to get through the second wave of COVID-19, and all of them are preparing to combat the possible third wave, which is expected to affect kids the most.
The exponential spread of COVID-19 not only puts a lot of pressure on the hospitals and treatment centres but also creates a huge burden on diagnostic labs.
The RT-PCR test is considered to be the sure-shot way of diagnosing COVID-19, and with a huge number of cases coming daily, the laboratories were finding it difficult to deliver test results even in 3-4 days. This was causing a delay in the hospitalisation of the patients.
With the introduction of these self-test kits, the COVID-19 management in India can see a huge improvement. It will release a lot of pressure from the limited manpower that has been responsible for collecting the samples, executing the tests and delivering the result.
Using these kits, people can check their status within just 15 minutes, enabling them to get the required treatment in time.
The United States was the first country to approve the use of such self-test kits back in November 2020. California-based Lucira Health had produced this all-in-one rapid result test kit and was approved for emergency use in the US. Europe and South Korea have also approved similar kits already.
There are certain shortcomings of these self-test kits as well. The biggest of them all is reliability. There are chances of improper sample collection or contamination of swab sticks.
There are high chances of rapid antigen tests showing false-negative results. In that case, a COVID-positive asymptomatic person may turn out to be a super spreader of the virus without even being traced.
On one hand, where the self-test kits can help in easing the pressure on diagnostic labs and hospitals, on the other, over-dependence on this particular method can lead to an even worse situation. Hence, it should only be recommended and used by those who can carry out the guidelines perfectly and interpret the result accurately.
Disclaimer: The above information has been prepared by a qualified medical professional and may not represent the practices followed universally. The suggestions listed in this article constitute relatively common advice given to patients and since every patient is different, you are advised to consult your physician, if in doubt, before acting upon this information. Lupin Limited has only facilitated the distribution of this information to you in the interest of patient education and welfare.

Leave your comment...
Comments