Sputnik V Gets Emergency Use Approval In India
By Dr. Nikita Toshi +2 more

Get,

to manage your symptom
Get your,


4 Cr+ families
benefitted

OTP sent to 9988776655



You’ve successfully subscribed to receive
doctor-approved tips on
Whatsapp

Get ready to feel your best.

Hi There,



Register to Avail the Offer
Send OTPBy continuing, you agree with our Privacy Policy and Terms and Conditions

Hi There,

Trusted by 4 crore+ families

OTP sent to 9988776655



You have unlocked 25% off on medicines




Code: NU25

By Dr. Nikita Toshi +2 more
The new COVID-19 strain has derailed the Indian healthcare infrastructure in no time, and our country, which was donating vaccines to the neighbouring nations a few weeks ago, is facing a shortage of the life-saving shots and almost every other facility required to fight this pandemic.
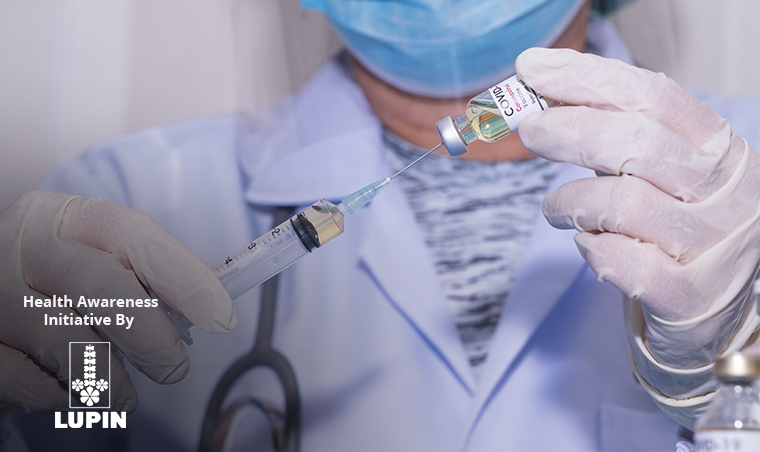
In a huge relief to the people, the Drug Controller General of India (DCGI) has approved the Sputnik V COVID-19 vaccine for emergency use in India. This is the third vaccine India has approved to use against the deadly virus. The earlier two are – Covishield and Covaxin. The former has been developed by the Oxford University-AstraZeneca and Pune-based Serum Institute of India (SII) is manufacturing it, while the latter has been developed and manufactured by Bharat Biotech and the National Institute of Virology (NIV).
Table of Contents
Indian pharmaceutical company Dr Reddy’s Laboratories has joined hands with the Russian sovereign fund Russian Direct Investment Fund (RDIF) for carrying out the bridge clinical trials of the vaccine in India, which is the 60th country to approve its use.
The RDIF has confirmed that vaccine distribution will begin by the end of April or in the first week of May.
The Gamaleya Research Institute of Epidemiology and Microbiology, backed by the Russian state, has developed the vaccine. The registration of the vaccine in Russia was done in August 2020 as Gam-COVID-Vac, and the “V” in the name of the vaccine stands for alphabet V. The developer has informed that the vaccine can be stored at 2-8 degree celsius.
Dr Reddy’s Laboratories has got the contract of distributing up to 250 million doses in India, by importing them. RDIF has also signed a manufacturing contract with Stelis Biopharma, Gland Pharma, Virchow Biotech, Panacea Biotec and Hetero Biopharma to manufacture more than 850 million Sputnik V doses in India. The doses produced in India will be exported across the world.
The production of the vaccine is soon to start at Hetero Biopharma and Panacea Biotec, while the other pharma companies will begin production in September 2021.
Sputnik V is an adenovirus viral vector vaccine, which carries viruses especially made containing the coronavirus genes. Some of these types of vaccines make their way into the cells and cause them to produce viral proteins. The others just gradually replicate, travelling through the body with virus proteins on their surface.
People have to get two doses of Sputnik V, similar to Covishield and Covaxin. Both the doses have to be administered at least 21 days apart. These are inoculated through injection into the muscle.
The Russian vaccine has shown excellent results with an efficacy rate of 91.5 percent. This rate is the highest after Moderna and Pfizer-BioNTech’s vaccines. Before giving it the green signal, a bridging trial was conducted in India.
Another positive news is that only 0.1 percent of the vaccine receivers have complained about the side effects.
In the international market, the Russian vaccine costs around Rs. 750 per dose, but its price in India is still to be decided, as the government is trying to negotiate the pricing with the manufacturer.
The vaccine approval for emergency use in India is very crucial as the second wave of the pandemic has already put India on the back foot in the war against the virus. Many states in the country have been reporting a shortage of vaccines, even when India is all set to expand the vaccination drive.
Russia became the first country to start inoculating its citizens with Sputnik V. It is currently being administered in many countries including Pakistan, Egypt, Argentina, Vietnam, Morocco, Bahrain, Jordan, Panama, Mauritius, Hungary, the United Arab Emirates, Iran, the Philippines and Sri Lanka.
With the quickly rising number of cases of COVID-19 in India, approval to Sputnik V for emergency use can come as a boon for the countrymen. We should hope that the first lot of vaccines land in India soon so that the vaccination drive can come back on the right track.
Know your current health status.

Disclaimer: The information provided here is for educational/awareness purposes only and is not intended to be a substitute for medical treatment by a healthcare professional and should not be relied upon to diagnose or treat any medical condition. The reader should consult a registered medical practitioner to determine the appropriateness of the information and before consuming any medication. PharmEasy does not provide any guarantee or warranty (express or implied) regarding the accuracy, adequacy, completeness, legality, reliability or usefulness of the information; and disclaims any liability arising thereof.
Links and product recommendations in the information provided here are advertisements of third-party products available on the website. PharmEasy does not make any representation on the accuracy or suitability of such products/services. Advertisements do not influence the editorial decisions or content. The information in this blog is subject to change without notice. The authors and administrators reserve the right to modify, add, or remove content without notification. It is your responsibility to review this disclaimer regularly for any changes.

Leave your comment...
Comments