Is Mounjaro Approved for Weight Loss? Here’s What Our Endocrinologist Says
By Dr. Akash N. Shah +2 more

Get,

to manage your symptom
Get your,


4 Cr+ families
benefitted

OTP sent to 9988776655



You’ve successfully subscribed to receive
doctor-approved tips on
Whatsapp

Get ready to feel your best.

Hi There,
Download the PharmEasy App now!!


Register to Avail the Offer
Send OTPBy continuing, you agree with our Privacy Policy and Terms and Conditions

Hi There,
Sign up on PharmEasy now!!
Trusted by 4 crore+ families

OTP sent to 9988776655



You have unlocked 25% off on medicines




Code: NU25
By Dr. Akash N. Shah +2 more
Table of Contents
Mounjaro (tirzepatide) is an injectable medication approved in 2024 by the Central Drugs Standard Control Organization (CDSCO) in India, for improving blood sugar management in adults with type 2 diabetes (T2DM), when used in conjunction with diet and exercise1,2. Since its approval, tirzepatide has earned significant attention not only for its glucose-lowering effects but also for its impact on weight management3,4.
This article explores the pharmacologic mechanisms of tirzepatide, reviews key clinical findings, examines emerging evidence on its effect on body weight and metabolic health, and considers expert opinions on the medicine.
Mounjaro (tirzepatide) is currently approved only for the treatment of T2DM. It is not officially approved for weight loss1.
However, in the United States, the Food and Drug Administration (FDA) has approved tirzepatide for weight loss under a different brand name known as Zepbound. This brand is specifically used for chronic weight management in adults with obesity or those who are overweight with a minimum of one weight-related condition (such as hypertension)5.
Therefore, to answer the question, “is Mounjaro approved for weight loss?”, while the brand Mounjaro is not approved for weight loss in India, Zepbound (another brand name for tirzepatide) is approved in the U.S. for weight management, although not specifically as a weight loss drug.
Tirzepatide works by stimulating two natural gut hormones: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide)6. These hormones help regulate blood sugar, insulin levels, and appetite7.
By activating both GLP-1 and GIP receptors, tirzepatide:
These combined effects may support better blood sugar control and may help in weight management. Therefore, for people wondering “how does Mounjaro help you lose weight,” the answer lies in its unique ability to influence both metabolic and appetite-related pathways, leading to reduced appetite and altered eating patterns over time.
Mounjaro is primarily prescribed for adults with T2DM, especially those with insulin resistance who need better blood sugar control9.
Mounjaro may also be considered for patients who have obesity or metabolic syndrome, where improving insulin sensitivity and managing weight is essential. Although Mounjaro is not officially approved for weight loss in India, weight management plays a key role in managing conditions like T2DM, high blood pressure, and cardiovascular risk9. In such cases, Mounjaro may be used strictly under medical supervision to support overall metabolic health and long-term disease management.
Mounjaro is given as an injection (once weekly), usually at the same time each week. It is injected under the skin (subcutaneously) at common injection sites including the stomach or thigh, or upper arm10.
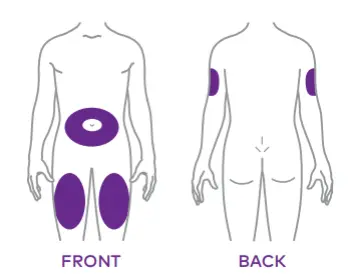
Note: It’s important to rotate injection sites each week to avoid irritation10,11.
While people often search for the “best place to inject Mounjaro for weight loss,” the injection site doesn’t influence the effectiveness of the medication as long as the site is rotated every week.
Although Mounjaro is not officially approved for weight loss in India, clinical trials have used specific dosing schedules that have had a significant effect on weight10. The correct “Mounjaro dose for weight loss” should only be determined by a doctor based on the patient’s individual health needs.
Additional Note: Always use Mounjaro under medical supervision to ensure safety and effectiveness.
Patients and clinical studies have reported several noticeable changes while using Mounjaro. Commonly observed effects include:
These changes may support improved portion control and less frequent cravings (or snacking), which may eventually support better weight and blood sugar management.
However, it’s important to note that these reported effects are not guaranteed. Responses to Mounjaro can vary based on individual factors like lifestyle, medical history, and how the body reacts to treatment. Always consult your doctor for any advice.
When using Mounjaro for diabetes management, some patients report side effects, especially during the first few weeks as the body adjusts. Commonly noted Mounjaro side effects include6,10:
Tips to manage the above gastrointestinal side effects of Mounjaro12:
While many people tolerate Mounjaro well, it’s important to monitor people taking Mounjaro for weight loss for side effects and consult a doctor to ensure safe and effective use.
In my experience, when taking Mounjaro, always consult a qualified dietitian or nutritionist to review your diet plan. This helps prevent gastrointestinal discomfort, Hypoglycemia-related dizziness, and other potential complications linked to unbalanced nutrition.
Dr. Malavika Athavale, Consulting Clinical and IBS Nutritionist
Clinical trials, including the SURMOUNT series, have explored the impact of tirzepatide (the active ingredient in Mounjaro) on weight management. In these studies, participants who received tirzepatide experienced significant weight reduction, depending on the dose and study duration6,13.
For example, in SURMOUNT-1, adults with obesity (without diabetes) lost up to 21% of their body weight over 72 weeks14.
In comparison to other GLP-1 medications, such as semaglutide, Mounjaro showed a greater average reduction in body weight in clinical trials15.
Important: These outcomes reflect clinical trial data and do not guarantee the same results for everyone. Weight changes depend on individual factors, including adherence to treatment, lifestyle, and overall health.
Also Read: Mounjaro vs Zepbound: Differences, Uses, Side Effects & More
In India, Mounjaro is available by prescription and is primarily indicated for T2DM. Its price starts from three thousand rupees and can vary based on the dosage and the pharmacy where it’s purchased. Some insurance plans or discount programs may help reduce the cost. It’s best to check with your local pharmacy or insurance provider for specific pricing and coverage details.
While many people search for terms like “Mounjaro price for weight loss” or “how to get Mounjaro for weight loss,” it’s important to note that the drug is not approved for weight loss in India and its use for weight management should be considered strictly under the guidance of a doctor.
Note: Patients are strongly advised to avoid sourcing Mounjaro online without a prescription, as it should only be used under medical supervision to ensure safety and effectiveness.
Before starting Mounjaro, it’s important to understand who it’s meant for and when it may not be appropriate:
Also Read: Mounjaro vs Ozempic: Our Endocrinologist Explains the Difference
Mounjaro is an FDA-approved medication for T2DM and, while not officially approved for weight loss in India, some patients have experienced additional metabolic benefits, including changes in appetite and weight.
However, it is not a substitute for healthy lifestyle choices. Long-term success in managing diabetes, weight, or related conditions depends on a combination of medical guidance, balanced nutrition, regular physical activity, and consistent follow-up.
Always consult a doctor before starting or continuing any medication like Mounjaro. Remember, safe, supervised use is key to achieving the best outcomes.
Also Read: Mounjaro (Injection): Uses, Side Effects, Dosage, and How It Works
No, Mounjaro (tirzepatide) is not approved for weight loss in India; its use is strictly limited to improving glycaemic control in adults with T2DM16.
In clinical trials where the effect of tirzepatide was studied in individuals with obesity over 72 weeks:
-People without diabetes lost up to 20.9% of their body weight (SURMOUNT-1)14.
-People with T2DM lost up to 14.7% (SURMOUNT-2) of body weight17.
Note: These results are based on trial data (individual outcomes may vary).
Mounjaro should be taken once a week as a subcutaneous injection (under the skin), typically in the stomach, thigh, or upper arm, with rotation of injection sites each time. It must be used under medical supervision, as your doctor will determine the appropriate dose and monitor for side effects10. Do not self-prescribe, especially for weight loss; safe use requires professional guidance.
In India, Mounjaro is approved only for T2DM16. Its use for weight loss in people without diabetes is not officially approved and should only be considered under medical supervision.
Disclaimer: The information provided here is for educational/awareness purposes only and is not intended to be a substitute for medical treatment by a healthcare professional and should not be relied upon to diagnose or treat any medical condition. The reader should consult a registered medical practitioner to determine the appropriateness of the information and before consuming any medication. PharmEasy does not provide any guarantee or warranty (express or implied) regarding the accuracy, adequacy, completeness, legality, reliability or usefulness of the information; and disclaims any liability arising thereof.
Comments

Leave your comment...
You may also like
Comments